BBS Automation partners with life science manufacturers to support discovery, development, industrialization, and manufacturing through automation engineered for regulatory readiness. We help customers reduce integration risk, improve repeatability, strengthen traceability, and scale production across highly regulated applications. From pilot and clinical builds to full production systems, BBS delivers automation solutions designed to support quality, precision, and speed to market.
Built for Compliance. Powered by Innovation.
Life science manufacturing demands more than throughput. It requires precise process control, repeatable performance, and the documentation discipline needed to support validation and audit readiness. BBS combines advanced automation with deep experience in regulated manufacturing so customers can move faster without compromising quality.
Why customers choose BBS
| Regulatory Readiness | Realiability and Precision | Innovation That Supports Commercialization | Partnership from Concept to Production |
|---|---|---|---|
| Evolving FDA, EMA, ISO, and cGMP expectations add complexity across product development and manufacturing. BBS systems are engineered to support traceability, documentation, quality assurance, and validation-focused execution across each process step. | Complex therapies and sensitive products require stable, repeatable automation. BBS platforms are designed to improve accuracy, reduce human error, minimize downtime, and support consistent, scalable production performance. | Advanced robotics, vision inspection, AI-assisted analytics, modular design, and digital twin methods help reduce risk during development and scale-up. The result is smoother industrialization and faster readiness for launch. | BBS works as a systems integration partner across the full lifecycle, from Proof of Principle through pilot, clinical, and full-scale production. We collaborate closely with customers to align automation strategy with process needs, capacity goals, and future growth. |
Protecting the Innovation Behind Your Product
In life sciences, automation is not only about performance. It is also about protecting the product knowledge, process expertise, and proprietary methods that create competitive advantage. BBS supports confidential customer programs through disciplined project governance, controlled collaboration, structured documentation practices, and respect for customer-owned intellectual property. That gives teams a trusted environment to advance sensitive products and processes while moving toward scalable manufacturing.
Confidential development support for high-value products, proprietary processes, and next-generation drug delivery platforms.
What BBS Brings to Complex Life Science Automation
| Item | Turnkey automation from Proof of Principle to high-volume production |
| Item | Precision assembly, testing, inspection, and traceability |
| Item | Digital twin methods and simulation-driven development |
| Item | Cleanroom-compatible system design |
| Item | Modular platforms built for scale and future upgrades |
| Item | Remote diagnostics and global lifecycle support |

Automation for Complex Life Science Products
BBS supports life science applications where precision, compliance, and scalable performance are critical.
• Injectable drug devices
• Combination products
• Medical devices
• Diagnostics and lab automation
• Drug delivery systems
• High-volume regulated consumables
From Ideation to Execution
Step 1: Assess Critical Requirements
We align early on product, process, compliance, throughput, and risk priorities.
Step 2: De-Risk the Automation Strategy
We use concepting, Proof of Principle support, modular design, and simulation methods to reduce uncertainty.
Step 3: Engineer for Scale
We build systems that support repeatable performance, validation activities, and future manufacturing growth.
Step 4: Support the Full Lifecycle
Our teams provide service, training, upgrades, diagnostics, and long-term optimization after installation.
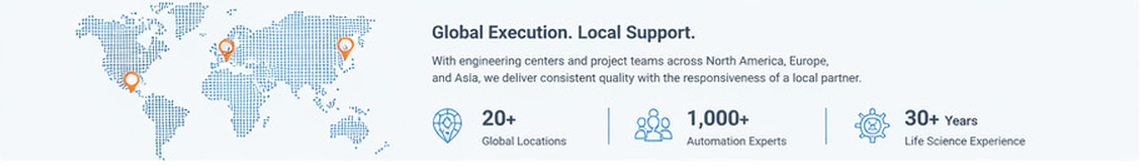
Global Execution. Local Support.
BBS Automation combines worldwide systems integration capability with responsive regional support. For North American customers, that means access to local collaboration backed by a global engineering organization experienced in regulated, high-precision manufacturing. It is a delivery model designed to reduce risk, improve communication, and support long-term success.
Ready to De-Risk Your Next Launch?
Meet BBS Automation at PFS Boston to discuss how validated, future-ready automation can support your next innovation in pharma, diagnostics, medical devices, or drug delivery systems.
Buttons
• Book a Meeting at PFS Boston
• Contact Our Life Sciences Team

Frequently Asked Questions
BBS Automation designs and integrates automation systems for regulated life science manufacturing, supporting applications in pharmaceuticals, medical devices, diagnostics, and drug delivery.
BBS supports automation from Proof of Principle and pilot builds through clinical and full-scale production.
BBS helps reduce risk through modular design, simulation methods, precision engineering, traceability, validation-focused execution, and lifecycle support.
Yes. BBS supports confidential customer programs through disciplined project governance, controlled collaboration, and structured documentation practices.





